FOOD SAFETY ARTICLES
Featured

Heavy Metals in Baby Food: Regulating Raw Ingredients to Ensure Food Safety
By Jaclyn Bowen
In recent years, the issue of heavy metal contamination in baby food has emerged as a pressing concern, prompting nationwide
Read More
The Path to Sustainable Food: EU Regulations and Cell Cultured Meat
By Mathilde Do Chi
As the world grapples with the need for sustainable food solutions, the development of products like cell cultured meat
Read More
Enhancing Global Food Safety Compliance Through ISO 22000
By Jacqueline Southee
The key responsibility of the food industry is to make, distribute, and deliver safe food, and there are standards to
Read More
Safeguarding Our Future: Where Food Safety and Sustainability Meet
By Anthony Raschke
As populations grow, resources deplete, and climate change accelerates, the need for innovative solutions to ensure the safety of our
Read More
Preserving Food Quality: The Critical Role of Packaging in Safeguarding Food Safety and Sustainability
By Sasha Matera-Vatnick
Consumers perceive value in transparency from food and beverage companies, and one area that has become top of mind for
Read More
Mitigating Risks to Food Safety from Climate Change
By Daphne Ewing-Chow
With 2023 being the hottest year on record, it is no surprise that foodborne illnesses have also been at an
Read More
Revolutionizing Food Safety: Insights from Dubai’s FoodWatch
Podcast Content Hosted and Produced by Global Food Safety Resource
In a world where food safety concerns are ever-present, innovative solutions are crucial
Read More
From UVC to Far-UV: Comparing the Efficacy, Safety, and Cost of UV Technologies for Pathogen Control
By James Rush
The food processing industry has long used ultraviolet (UV) light as an effective mechanism for pathogen deactivation. Ultraviolet-C (UVC), also
Read More
Innovating Against Waste: Using Technology to Reduce Food Waste
Content Produced by GFSR
Food waste is a paradox of our times: while millions go hungry, vast amounts of food are lost or
Read More
Sanitation Symbiosis: Winning the War Against Pests in Food Processing Facilities
By Zia Siddiqi
Food processing facilities play a crucial role in ensuring the safety and quality of the food supply chain, especially
Read More
The Recipe for Responsibility: Ensuring Allergen-Free Dining Experiences
By Robert McKeon
The food service industry is facing an increasing number of inquiries from customers regarding food allergens. This growing concern mandates
Read More
2024 Food Safety Forecast: Embracing Transparency and Advanced Technology
By Andrea Tolu
From year to year, the food safety landscape is fast moving and ever changing, so it’s a challenge to know
Read More
Sanitation Excellence: Proven Tips for Impactful Food Safety Programs
By Sean Berdan
Once, not so long ago, the primary aim of a sanitation program was to prevent a facility from emitting unpleasant
Read More
Beyond Recall: The Cost of Food Safety Failures and the Value of Preventive Controls
In the fast-paced world of food manufacturing, ensuring the safety of the products that reach consumers is critical. Food safety incidents not
Read More
The Perils of PowerPoint: New Ways to Boost Engagement in Food Safety Training
By Lute Atieh
In the world of food safety training, the battle against boredom is real, and PowerPoint presentations are the sworn
Read More
Three Things You Can Do Today to Empower Your People and Improve Food Safety
By Alex Hanley
When was the last time you took a walk through your facility and had a simple conversation with someone on
Read More
The Vital Role of Non-Food Suppliers in Food Fraud Prevention
By John Spink
If you sell products to the food industry, then you ARE in the food industry and ARE required to
Read More
What You Need to Know About the USDA’s Enhanced Organic Enforcement Rule
By Allison McLeod
Year over year, consumers seek more nutritious and healthier food options when grocery shopping. A study from the Read More

Training with Impact: 4 Ways to Guarantee Consistent Execution in Your Plant
By Alex Hanley
In the food industry, training is a means to an end, and the end is consistent performance in the plant.
Read More
‘Poisoned’ vs. Progress: A Balanced View on Food Production and Regulation
BLOG POST
By Keith Warriner
For many consumers, their only concern around food is how to navigate the aisles of the supermarket
Read More
Beyond the Binders: The Future of Tech-Enhanced Food Safety Audits
By Tyler Williams
Imagine walking through a manufacturing facility, virtually witnessing an audit through a pair of smart glasses.
Next, imagine using
Read More
INDUSTRY STANDARDS AND REGULATIONS

Heavy Metals in Baby Food: Regulating Raw Ingredients to Ensure Food Safety
By Jaclyn Bowen
In recent years, the issue of heavy metal contamination in baby food has emerged as a pressing concern, prompting nationwide
Read More
The Path to Sustainable Food: EU Regulations and Cell Cultured Meat
By Mathilde Do Chi
As the world grapples with the need for sustainable food solutions, the development of products like cell cultured meat
Read More
Enhancing Global Food Safety Compliance Through ISO 22000
By Jacqueline Southee
The key responsibility of the food industry is to make, distribute, and deliver safe food, and there are standards to
Read More
Preserving Food Quality: The Critical Role of Packaging in Safeguarding Food Safety and Sustainability
By Sasha Matera-Vatnick
Consumers perceive value in transparency from food and beverage companies, and one area that has become top of mind for
Read More
Innovating Against Waste: Using Technology to Reduce Food Waste
Content Produced by GFSR
Food waste is a paradox of our times: while millions go hungry, vast amounts of food are lost or
Read More
2024 Food Safety Forecast: Embracing Transparency and Advanced Technology
By Andrea Tolu
From year to year, the food safety landscape is fast moving and ever changing, so it’s a challenge to know
Read More
What You Need to Know About the USDA’s Enhanced Organic Enforcement Rule
By Allison McLeod
Year over year, consumers seek more nutritious and healthier food options when grocery shopping. A study from the Read More

Bolstering Your ESG Efforts with Sustainability-Based Certifications
By Lisa Spicka
The food industry is one of the industries most impacted by climate change. Natural disasters and shifting temperatures impact growing
Read More
Expanding Edibles into Canada: Key Considerations for US Cannabis Companies
By Tina Chang
On October 17, 2018, the Government of Canada legalized and strictly regulated the production, distribution, sale, import, export, and possession
Read More
Get Your Data Together to Better Manage High-Risk Foods
By Angela Fernandez
The U.S. Food and Drug Administration’s (FDA) final rule on requirements for on requirements for additional traceability records for certain
Read More
FDA Guidance on Lead Contamination in Baby Food
By Jaclyn Bowen
The US baby food and infant formula industry has seen its fair share of food safety related headlines in
Read More
Is Sustainable Packaging Food Safe?
By Maria Spinetta
The latest trend in packaging is sustainability, but does it come at the cost of contaminating the food it is
Read More
Certifying to ISO 22003-1
Harmonization: ISO 22003-1 and 22003-2 for food safety audit and certification.
ISO 22003-1 is a supplement to ISO 17021-1: 2015, an
Read More
Put Science and Regulatory Insight Upfront for Meat Alternatives
By Annina Halvorseth and Annie-Laure Robin
As the market for meat alternatives continues to expand, plant-based products are an innovation hotspot. There’s
Read More
How to Prepare for the Baby Food Safety Act
By Jackie Bowen
In February 2021, the results of a congressional investigation revealed levels of arsenic, lead and other toxic metals
Read More
Where’s the Beef? Cell-Cultured Meat Grown in a Lab Needs Regulation
By Jayne Roth
What will we find next on the grocery store shelf? A plethora of food and beverages beyond your imagination continue
Read More
Nanocomposite Food Packaging is Not Ready for Prime Time
by Suzanne Osborne
Packaging improves food safety. It extends shelf-life, protects against damage, and limits pathogen contamination. Plastic is the most common packaging
Read More
Beware of Food Fraud During the Coronavirus
By Mitchell Weinberg
Crises of an environmental nature, or as in the case of Coronavirus, a pandemic, bring out the best and worst
Read More
Reducing the Threat of SARS-CoV-2: From Farm to Fork
By Keith Warriner
CoVid-19 is the most serious pandemic since the Spanish Flu of 1918. During this time the food industry has been
Read More
How Retailers and Producers Can Reduce Food Waste Safely and Sustainably
By Louise Grinyer
Approximately two thirds of UK consumers surveyed highlighted a willingness to compromise factors such as product consistency, availability and convenience
Read More
Virtual Reality Set to Modernize Food Safety Training
By Suzanne Osborne
The Food Safety Modernization Act has raised the standard for global food safety. As a consequence, there is a pressing
Read More
PREVENTATIVE CONTROLS AND RISK

Sanitation Symbiosis: Winning the War Against Pests in Food Processing Facilities
By Zia Siddiqi
Food processing facilities play a crucial role in ensuring the safety and quality of the food supply chain, especially
Read More
The Recipe for Responsibility: Ensuring Allergen-Free Dining Experiences
By Robert McKeon
The food service industry is facing an increasing number of inquiries from customers regarding food allergens. This growing concern mandates
Read More
Sanitation Excellence: Proven Tips for Impactful Food Safety Programs
By Sean Berdan
Once, not so long ago, the primary aim of a sanitation program was to prevent a facility from emitting unpleasant
Read More
Beyond Recall: The Cost of Food Safety Failures and the Value of Preventive Controls
In the fast-paced world of food manufacturing, ensuring the safety of the products that reach consumers is critical. Food safety incidents not
Read More
How Global Events are Reshaping Food Safety Strategies
By Andrea Tolu
When the Covid-19 pandemic hit in 2020—in the words of the OECD, the Organisation for Economic Co-operation and Development—
Read More
Communication: Your Cyber Crime Fighting Secret
By Linda Eatherton
Cybercrime threatens every operating organization. Data thieves and trolls lurk in cyberspace waiting for an opportunity to attack, and
Read More
The Importance of AI and Predictive Analysis in Food Safety Management Systems
By Maria-Eleni Dimitrakopoulou
In the 21st-century food safety challenges remain a hurdle since incidents all over the world are constantly reported. Foodborne pathogens,
Read More
When Risks Evolve: How to Manage Emerging Foodborne Pathogens
By Richard Onley
Most food businesses have longstanding strategies in place to monitor and manage microbiological safety. Yet food safety risks are continually
Read More
An Ounce of Prevention: Comparing US and Canadian Food Safety Regulations
By Nidhi Chaturvedi
The goal of both the Safe Food for Canadians Regulations (SFCR) and the Food Safety Modernization Act (FSMA) is
Read More
How Poor Pest Management Can Affect Your Food Safety
By Violetta Njunina
Pests destroy crops, contaminate food, and spread disease through their fur, droppings, saliva, urine, and bacteria. As such, they
Read More
3 Preventative Controls of Pathogens on Low Moisture Foods
By Keith Warriner
My previous article on low moisture foods provided a background into the food safety issues and pathogens linked to low
Read More
Take Steps Now to Protect Your Company from a Cybersecurity Attack
By Faith Beaty
Food safety, information and cyber security may not seem to be related topics, nevertheless cyber threats are risks within the
Read More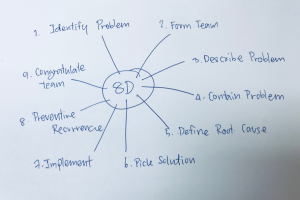
Demystifying Root Cause Analysis: Key Steps to Approach Problem Solving
By Geoff Farrell
Root Cause Analysis (RCA) is a systematic method of problem solving that can be used to identify and resolve core
Read More
Know Your Limits: How Pesticides Protect Your Food Supply
By Dilia Narduzzi
Before a new pesticide is brought to market, it undergoes rigorous testing to make sure that handling it is
Read More
Climate Issues: Drastic Change Needed to Protect Food Safety and Security
By Dr. Louise Manning
Consumers expect the food they purchase to be high-quality and safe. Assuring food security means there must be sufficient,
Read More
Managing Risk Will Ensure Food Safety Post-Pandemic
By Dr. Sylvain Charlebois
The COVID-19 pandemic has brought a new era of food safety, in which risks are managed and perceived
Read More
Protect Your Brands From Expiry Date Fraud
By Karen Constable
Food fraud takes many forms, but it primarily falls into two categories. Either the food itself is adulterated, or
Read More
Consumer Trust Can Be Earned Even in a Pandemic, Here’s How
By Lisa Jo Lupo
Striving for perfection is an ideal goal in business. But in doing so, it is essential to face the
Read More
How to Protect Your Company from Organic Food Fraud
By Karen Constable
In the first part of this two-part series, we explained the increased number of cases of food fraud in
Read More
The Most Unexpected Way to Commit Food Fraud? Organics.
By Karen Constable
With growing consumer demand, a premium price and perceived health benefits, organic food sits squarely in the sights of food
Read More
How to Manage Risks in the Food Industry Post-COVID-19
By Brita Ball
The pandemic has led to thousands of essential food workers becoming ill or asymptomatic carriers of the virus. Many
Read More
FOOD SAFETY COMPLIANCE

The Path to Sustainable Food: EU Regulations and Cell Cultured Meat
By Mathilde Do Chi
As the world grapples with the need for sustainable food solutions, the development of products like cell cultured meat
Read More
Enhancing Global Food Safety Compliance Through ISO 22000
By Jacqueline Southee
The key responsibility of the food industry is to make, distribute, and deliver safe food, and there are standards to
Read More
The Recipe for Responsibility: Ensuring Allergen-Free Dining Experiences
By Robert McKeon
The food service industry is facing an increasing number of inquiries from customers regarding food allergens. This growing concern mandates
Read More
What You Need to Know About the USDA’s Enhanced Organic Enforcement Rule
By Allison McLeod
Year over year, consumers seek more nutritious and healthier food options when grocery shopping. A study from the Read More

In the Driver’s Seat: Five Ways to Elevate Your Audit Experience
By Alex Hanley
Food safety audits, when done right, can be both fair and effective – but in order to optimize the experience
Read More
Fortifying the Future: Food Safety and Business Resilience
Content Produced by GFSR
Every food and beverage organization wants to be known for consistently delivering safe, high-quality food products to consumers
Read More
In Brief: The WHO’s Global Strategy for Food Safety through 2030
By Mariella Bustamante
At the 75th Session of the World Health Assembly held in May 2022, the World Health Organization (WHO) proposed a
Read More
Involved in a Food Safety Dispute? Time to Call an Expert Witness
By Andrea Tolu
When food quality and safety disputes happen, the parties involved may not always agree on who’s right or wrong.
Read More
How Gamification, Computer Vision and IoT is Improving Hand Hygiene Compliance
By Andrea Tolu
Hand hygiene is a long-standing issue in the food industry. While hand hygiene may seem to be simple and
Read More
Preparing for an FDA Inspection a Necessary Part of Your Food Safety Plan
By Kathy Knutson
Are you worried about the FDA showing up on your doorstep? Even the most diligent companies wonder when the FDA
Read More
Dissolvable Packaging Requires Regulatory Insight
By Dr Caroline Potter
Dissolvable packaging for food and beverage has emerged as a possible solution to some of the most challenging issues
Read More
Lessons from the Cannabis Industry
By Dyanne Parnel
With the emergence of the Covid-19 pandemic, manufacturers of consumer goods across the globe are
Read More
How to be Audit Ready
By Baharak Chegeni
Being “audit ready” is a hot topic in the food safety industry, as any expert would agree that audits are
Read More
Effectively Digitizing Food Safety Audits
By Judy Sebastian
The age of paper-based auditing and reporting is gradually coming to an end. More organizations and even municipalities are
Read More
Dubai Leadership: The Digitization of Food Safety Management
By John G. Keogh
“Food safety management will go completely digital by the end of 2020” according to Jehaina Al Ali from the
Read More
Food and Cannabis Industries to Pay Heed to the Good Manufacturing Practices
By Kathy Knutson
The cannabis industry is currently experiencing explosive growth, both in the United States and internationally. Yet the current landscape is
Read More
Auditor Shortage Slowly Being Addressed
Posted: Tuesday, October 15, 2019
By Lois Harris
Better use of technology could help ease the labour shortage in the auditing field according to
Read More
Using Innovative Technology to Solve Historic Food Safety Challenges
By Jacki Ross
For decades, the entire food industry has complied with cross-border U.S. Food and Drug Administration (FDA), and health and safety
Read More
Temperature Control: Affordable, Real-time Data Recorders Finally Available
By Bryan Smith
There are many reasons why controlling the storage environment during food transportation is important, not
Read More
Food Safety and Shifting Global Pressures: Are We Ready? Part 2
Posted: Tuesday, August 14 2019 In the first part of this two-part series on Food Safety and Shifting Global Pressures, we
Read More
Give Your Customers The Novel Foods They Want – Safely
By Margaret Spence Krewen
With the globalization of the food supply and consumer demand for more global food sources, there has been a
Read More
FOOD SAFETY CULTURE AND TRAINING

Sanitation Excellence: Proven Tips for Impactful Food Safety Programs
By Sean Berdan
Once, not so long ago, the primary aim of a sanitation program was to prevent a facility from emitting unpleasant
Read More
The Perils of PowerPoint: New Ways to Boost Engagement in Food Safety Training
By Lute Atieh
In the world of food safety training, the battle against boredom is real, and PowerPoint presentations are the sworn
Read More
Three Things You Can Do Today to Empower Your People and Improve Food Safety
By Alex Hanley
When was the last time you took a walk through your facility and had a simple conversation with someone on
Read More
Communicating Food Safety: A Part of Building and Protecting Your Brand
By Linda Eatherton
Nothing is more aggravating or concerning than to see misinformation spread about food safety protocols and technology. Distorted information can
Read More
Cybersecurity – a vital part of your company’s safety culture
By Kristin Demoranville
Incorporating safety programs into an organization’s culture is paramount in the food industry, where the integrity of the food supply
Read More
Getting Your Employees to Give a %$&# About Food Safety Training
By Lute Atieh
If your employees are reading Standard Operating Procedures and enduring death by PowerPoint for their new hire training or,
Read More
Cultural Intelligence Helps Build a Food Safety Culture
By Judy Sebastian
Today, my breakfast was the sum of fifteen ingredients coming together. In case you’re wondering, raisins were not one of
Read More
The New Normal for Workers and Food Service
By Lauren Solar
A post-COVID world still seems like an unrealistic dream, but before long we will emerge from the pandemic. How have these
Read More
3 Steps to Kickstart Your Food Safety Culture Program
By Austin Welch
Famed management scholar Peter Druker once claimed, “Culture eats strategy for breakfast.” While strategy is important, company culture ultimately drives
Read More
Determining Your Return on Investment (ROI) for Food Safety Training
By Akhila Vasan
Do you remember the last time you started a new job? All the hours of onboarding. Maybe some basic
Read More
How Memes and Millennial Culture Can Keep Food Safe
By Prof. Dr. Darin Detwiler.
I recently heard a joke that a Zoom call is much like holding a séance – the older
Read More
Building Quality Management into Food Safety Training Can Help Mitigate Your Company’s Risk
By Michelle Rzendzian
When it comes to training and food safety, quality management is key in ensuring consistency and excellence. An organization
Read More
The Psychological Secret to Employee Motivation Will Improve Food Safety
By Austin Welch
It’s an early spring morning on March 21, 2007, in the small town of Blakely, GA, and the quality manager
Read More
How Encouraging Psychological Safety Prevents Disaster
By Austin Welch
In March of 1977, the small airport on the island of Tenerife in the Canary Islands was cloaked in heavy
Read More
How to Make the Most of Your Virtual Training
By Tina Brillinger
Food safety training is no longer confined to webinar platforms. With the advent of Covid-19 and social isolation, companies have
Read More
How to Blow the Whistle on Food Safety Issues
By Judy Sebastian
The impact of whistleblowing is a long-lasting one. By definition, a whistleblower is an individual who “reveals something covert or
Read More
Key to Staff Retention in Restaurants is to Implement a Food Safety Culture
By Lauren Solar
The food service sector has a high staff turnover rate, with industry estimates suggesting that fast food workers leave at
Read More
Protecting Essential Workers in the Food Industry Necessary
By Susie Hoeller
March 2020 was the onset of millions of hospitalizations and deaths, as well as the unprecedented economic and social
Read More
The Key to Maintaining Food Safety during the Pandemic is Clear Communication
By Judy Sebastian
As 2020 unfurled, so did the optimism, confidence and motivation of people around the world. Leaders from various countries were
Read More
Empathy Key to Building a Strong Food Safety Culture
By Joan Martino
Is employee behaviour aligned with better food safety outcomes?
Unquestionably the answer is, “yes!” but it comes packaged along with
Read More
Energize Your Brand With A Strong Food Safety Culture
By Brita Ball
What if “food safety” were part of your company’s brand and your employees consistently reinforced that message through their behaviour?
Read More
FOOD SCIENCE AND TECHNOLOGY

Heavy Metals in Baby Food: Regulating Raw Ingredients to Ensure Food Safety
By Jaclyn Bowen
In recent years, the issue of heavy metal contamination in baby food has emerged as a pressing concern, prompting nationwide
Read More
The Path to Sustainable Food: EU Regulations and Cell Cultured Meat
By Mathilde Do Chi
As the world grapples with the need for sustainable food solutions, the development of products like cell cultured meat
Read More
Enhancing Global Food Safety Compliance Through ISO 22000
By Jacqueline Southee
The key responsibility of the food industry is to make, distribute, and deliver safe food, and there are standards to
Read More
Safeguarding Our Future: Where Food Safety and Sustainability Meet
By Anthony Raschke
As populations grow, resources deplete, and climate change accelerates, the need for innovative solutions to ensure the safety of our
Read More
Preserving Food Quality: The Critical Role of Packaging in Safeguarding Food Safety and Sustainability
By Sasha Matera-Vatnick
Consumers perceive value in transparency from food and beverage companies, and one area that has become top of mind for
Read More
Mitigating Risks to Food Safety from Climate Change
By Daphne Ewing-Chow
With 2023 being the hottest year on record, it is no surprise that foodborne illnesses have also been at an
Read More
Revolutionizing Food Safety: Insights from Dubai’s FoodWatch
Podcast Content Hosted and Produced by Global Food Safety Resource
In a world where food safety concerns are ever-present, innovative solutions are crucial
Read More
Innovating Against Waste: Using Technology to Reduce Food Waste
Content Produced by GFSR
Food waste is a paradox of our times: while millions go hungry, vast amounts of food are lost or
Read More
Sowing the Seeds of Food Safety: Mitigating Risks in Agriculture
Content Produced by GFSR
In today’s increasingly interconnected global food system, agricultural practices lay the foundational stone for overall food safety. With
Read More
The Future of Food is Two-Dimensional
By Liz Sertl
Digital technology has changed the way consumers engage with brands, retailers and information. We now expect easy access to an
Read More
How Global Events are Reshaping Food Safety Strategies
By Andrea Tolu
When the Covid-19 pandemic hit in 2020—in the words of the OECD, the Organisation for Economic Co-operation and Development—
Read More
Fortifying the Future: Food Safety and Business Resilience
Content Produced by GFSR
Every food and beverage organization wants to be known for consistently delivering safe, high-quality food products to consumers
Read More
Building the Case to Digitize Your Food Safety Testing
By David Hatch
We hear a lot about the efficacy of food safety testing. The benefits of having a well-rounded and robust testing
Read More
Catastrophic Failure: Lessons Learned from the Top 5 Food Safety Events
Content Produced by GFSR
Over the past 25 years, the food industry has witnessed several food safety incidents that have had severe consequences
Read More
Communicating Food Safety: A Part of Building and Protecting Your Brand
By Linda Eatherton
Nothing is more aggravating or concerning than to see misinformation spread about food safety protocols and technology. Distorted information can
Read More
Supplement Safety Regulations: Food vs. Pharmaceuticals
By Dilia Narduzzi
Supplements are big business. According to Grandview Research, the 2021 global dietary supplement market was valued at over 150 billion
Read More
How the Internet of Things (IoT) Improves Food Traceability
By Emily Newton
Internet of Things (IoT) technology has helped people gain visibility over everything from patient vital signs to the environmental characteristics
Read More
Cybersecurity – a vital part of your company’s safety culture
By Kristin Demoranville
Incorporating safety programs into an organization’s culture is paramount in the food industry, where the integrity of the food supply
Read More
The Importance of AI and Predictive Analysis in Food Safety Management Systems
By Maria-Eleni Dimitrakopoulou
In the 21st-century food safety challenges remain a hurdle since incidents all over the world are constantly reported. Foodborne pathogens,
Read More
CleanTech 2 – A green solution revolutionizing sanitation in the food industry
By Anselm Doering
Since the pandemic, there has been a heightened concern and awareness surrounding the uses and controls of pathogens, sanitizers
Read More
Top Food Safety Trends to Keep Watch for in 2023
By Chris Koger
Headlines from 2022 are influencing some of the food safety tech trends in store for this year, as the Food
Read More
FOOD SAFETY CONFERENCES
- IAFP's European Symposium on Food Safety
- April 30, 2024 - May 2, 2024
- Food Safety Summit
- May 6, 2024 - May 9, 2024
- IAFP Annual Meeting 2024
- July 14, 2024 - July 17, 2024
- Ontario Food Processing Association (OFPA) Annual General Meeting
- September 30, 2024
- all events





